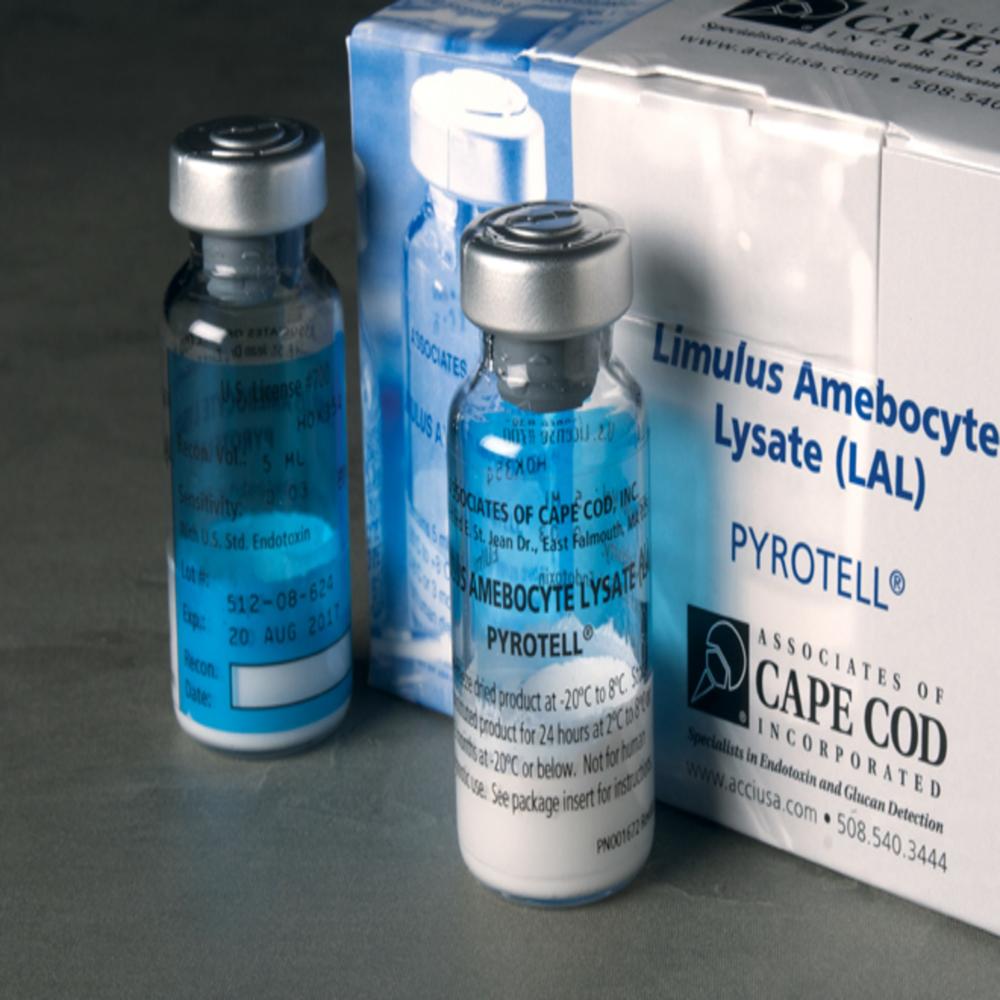
Bacterial Endotoxin Lal Test Kit
Bacterial Endotoxin Lal Test Kit Specification
- Product Type
- Bacterial Endotoxin LAL Test Kit
- Use
- Testing bacterial endotoxins for quality control
- Automation Grade
- Manual
- Suitable For
- Laboratories Pharmaceutical industries
- Properties
- High sensitivity, rapid results, validated specificity
- Application
- Detection of bacterial endotoxins in pharmaceutical and clinical samples
- Height
- 6 cm (kit box)
- Color Code
- Blue and White packaging
- Temperature Range
- 2C to 8C, avoid freezing
- Shape
- Rectangular box
- Accuracy
- 0.01 EU/ml detection limit
- Material
- Assay kit with reaction vials
- Weight
- ~450g (complete kit)
- Function
- In vitro endotoxin detection
- Dimension (L*W*H)
- Individual vials and full kit box packaging, approx. 20x12x6 cm
- Humidity Range
- Store below 70% RH
- Treatment Type
- Microbiological/Endotoxin testing
- Phase
- Lyophilized/Reagent
- Storage Capacity
- Multiple test vials per kit
- Sample Types
- Pharmaceutical water, medications, biologicals
- Shelf Life
- 12-18 months under recommended conditions
- Kit Contents
- LAL reagent, control standard endotoxin, reaction tubes, instructions
- Sensitivity
- As per pharmacopeial standards
- Test Method
- Limulus Amebocyte Lysate (LAL) Gel Clot
About Bacterial Endotoxin Lal Test Kit
Bacterial Endotoxin Lal Test Kit
As a prestigious name in this sector, we are persistently indulged in designing and fabricating the best in class Bacterial Endotoxin Lal Test Kit for our honored clients. This kit is ideal for measuring the amount of endotoxin in a protein, peptide and antibody sample by using the Limulus amebocyte lysate assay. It is sensitive, fast and accurate. In addition to this, our offered Bacterial Endotoxin Lal Test Kit measures the pierce LAL chromogenic endotoxin quantitation kit.
Features:
- Easy to use
- Accurate result
- Perfect finish
Technical Specifications
| Vial Contains | 2 ML |
| Injectable Form | Lyophilized Powder |
| Sensitivity | 0.03 AND 0.125 EU/mL |
| Result Type | Qualitative |
| Shelf Life | 24 Months |
Precision Endotoxin Detection
The LAL Gel Clot Test Kit provides reliable detection of bacterial endotoxins in pharmaceutical and clinical samples with exceptional sensitivity and specificity. Its validated process meets pharmacopeial standards, ensuring compliance for quality control purposes across various laboratory settings.
Comprehensive Kit Contents
Each kit includes everything required for testing, such as lyophilized LAL reagent, control standard endotoxin, reaction tubes, and clear user instructions. The blue and white packaged box is lightweight and compact, making it convenient for laboratory use and storage.
Optimized Performance and Storage
The test kit is designed to deliver rapid and accurate results. Storage conditions are critical: keep the kit between 2C and 8C and below 70% relative humidity to maintain reagent stability through its 1218 month shelf life. Avoid freezing the kit to ensure consistent performance.
FAQs of Bacterial Endotoxin Lal Test Kit:
Q: How does the Bacterial Endotoxin LAL Test Kit work?
A: The kit utilizes the Limulus Amebocyte Lysate Gel Clot method, which detects bacterial endotoxins by forming a gel clot in the presence of endotoxin. The result is observable and adheres to pharmacopeial standards for sensitivity and specificity.Q: What types of samples can be tested using this kit?
A: This kit is suitable for analyzing pharmaceutical water, medications, and biological samples. It is designed for in vitro endotoxin testing applications in laboratory and pharmaceutical industrial settings.Q: When should this test kit be used in quality control procedures?
A: Use the LAL Test Kit during routine quality control to ensure that pharmaceutical products and water are free from harmful levels of bacterial endotoxins, supporting compliance with regulatory safety requirements.Q: Where should the kit be stored to maintain its shelf life and accuracy?
A: Store the kit at temperatures between 2C and 8C and ensure humidity remains below 70% RH. Avoid freezing, as this may compromise reagent efficacy. Proper storage maintains kit sensitivity and a shelf life of 1218 months.Q: What is the process for performing an endotoxin test with this kit?
A: Follow the instructions provided in the kit. Add the LAL reagent and control standard endotoxin to your sample in the reaction tubes, incubate under recommended conditions, and observe gel clot formation to determine endotoxin presence.Q: What are the benefits of using this LAL Test Kit for endotoxin detection?
A: The kit offers high sensitivity, rapid results, and validated specificity, ensuring accurate endotoxin detection in a variety of sample types. It supports regulatory compliance and enhances safety in pharmaceutical quality control.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Bacterial Endotoxin Lal Test Kit Category
Endotoxin Lal Test Kit
Application : Detection of endotoxins in pharmaceuticals, medical devices, water, biological products
Use : Quantitative and qualitative detection of endotoxin
Product Type : Endotoxin LAL Test Kit
LYSATE 0.125
Price 5600.0 INR / Unit
Minimum Order Quantity : 1 Piece
Application : Essential for ensuring injectable drugs, medical devices, and raw materials are free from harmful bacterial endotoxins.
Use : Essential for ensuring injectable drugs, medical devices, and raw materials are free from harmful bacterial endotoxins.
Equipment Type : Limulus Amebocyte Lysate (LAL) Reagent with a specific sensitivity of 0.125 EU/mL